|
11/21/2023 0 Comments Ultibro lama labaThe Seebri Breezhaler treatment arms of the study were underpowered due to sample size. The study enrolled a total of 4,389 symptomatic, non-frequently exacerbating (up to one exacerbation in the previous year) patients with moderate COPD and 2,159 patients received Ultibro Breezhaler or continued their baseline therapy. Patients were recruited into four groups according to previous medication and symptoms, and randomized to a direct switch to Seebri Breezhaler (glycopyrronium) 50 mcg or Ultibro Breezhaler (indacaterol/glycopyrronium) 110/50 mcg once daily vs. Ultibro Breezhaler was also well tolerated in the CRYSTAL study, which was a prospective, multicenter, 12-week, randomized, pragmatic, open-label trial. “By showing that improved symptom control can be achieved through using Ultibro Breezhaler, the CRYSTAL study provides further support to limit the use of inhaled steroid-containing therapies to specific patient types that really need it.†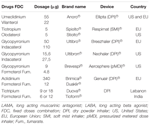 “Today’s results are significant as they show for the first time the positive effect of directly switching to Ultibro Breezhaler from other COPD treatments, such as inhaled steroid-containing combination therapies,†said Vasant Narasimhan, Global Head Drug Development and Chief Medical Officer for Novartis. GLYCOPYRRONIUM WITH INDACATEROL (Ultibro Breezhaler).  Significantly, CRYSTAL is the first LABA/LAMA pragmatic trial, designed to mimic clinical practice, so treatment switching occurred without a washout period. Combination Bronchodilator Preparations for COPD: LAMA With LABA. In the CRYSTAL study, patients with moderate COPD who were switched to Ultibro Breezhaler from their previous therapy (LABA+ICS or LABA or LAMA) experienced superior improvements in lung function (trough FEV 1) and breathlessness at week 12. Ultibro Breezhaler 85/43 1 dose OD 55/22 Forceful and deep Anoro Ellipta 1 dose OD Forceful and deep Spiolto Respimat 2.5/2. LABA/ LAMA combo Duaklir® Genuair® DPI Aclidinium/ Formoterol 340/12 mcg/puff COPD only 1 puff BD 60 No £32.50 COPD only Ultibro Breezhaler® DPI Glycopyrronium/i ndacaterol 85/43mcg/puff COPD only 1 puff od 30 NO £32.50 COPD ONLY. Novartis has released positive results from the first large-scale study exploring the effects of directly switching symptomatic, non-frequently exacerbating patients with moderate COPD from their current treatments, including steroid-containing combinations and long-acting bronchodilators, to the dual bronchodilator Ultibro Breezhaler (indacaterol/glycopyrronium) 110/50 mcg. The study, known as Claim, is the first to examine the effect of a med from the LABA/LAMA class on cardiac function. LABA + LAMA Prescribe Triple therapy STEP 5: REVIEW Prescribe Triple therapy Phenotype 1 STEP 1: ASSESSMENT STEP 2: DIAGNOSIS Post-bronchodilator FEV1/FVC ratio
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
